Cas No: 100-42-5
EINESC No: 202-851-5
Molecular weight: 104,15 g/mol
Chemical Formula: C8H8
Chemical Structure:
Physical Properties
General Properties: colorless, oil-like liquid
Odor: strawverry-like, sweet
Intensity: 0.909 g/cm3
Boiling point: 145 °C
Melting point: −30 °C
Flash point: 31 °C
Vapor pressure: 5 mmHg (20 °C)
Refraction index: 1.5469 nD
Solubility (aqueous) % 0.03 (20 °C)
Viscosity:
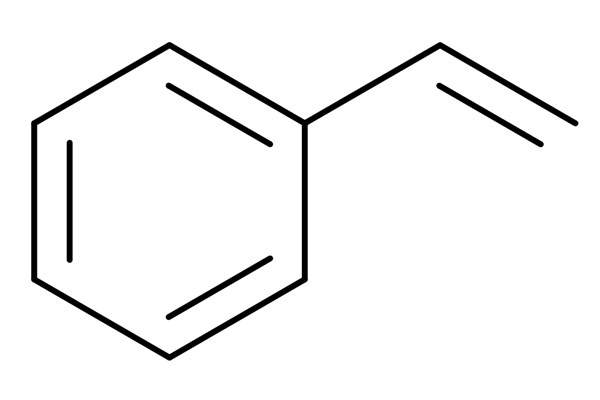
General Properties
Styrene, also known as ethylene benzene or vinyl benzene, is an organic compound which is a variant of benzene. Its odor is pleasant in low concentrations but disturbing in high concentrations. It is a pleasant scented, colorless aliphatic liquid that easily vaporized. Styrene, which is very suitable to polymerisation, is a pioneer to polysyrene and certain copolymers. It triggers polymerisation by itself but this reaction is exothermic and it can create explosions. Stolt Groenland Incident, which happened in South Korea in 2019, is caused by styrene autopolymerisation.
Production
Styrene is industrially produced from ethylene benzene in high amounts. Almost all ethylene benzene production is for styrene.
There are two preferred methods. In first, one hydrogen of ethylene benzene is severed via vaporization which is catalyzed by iron oxide. This process is 88-94% fruitful and reversible. Then, styrene is removed from ethylene benzene via distillation. Polymeriztion inhibitor must be used to prevent autopolymerization.
In second, ethylene benzene is given oxygen to create hydroperoxide. Then, it is used to oxidise propylene to propylene oxide. 1 remaining phenylethanol is dehydrated to acquire styrene.
It can also be produced by toluen, methanol, benzene and ethane but they are not as preferable as aforementioned methods.
Applications
Styrene is easily polymerized as it contains vinyl group. It is used in production of polymers that are used in polystyrene, ABS, SBR rubber, SBR latex, SIS, SAN and unsaturated polyesters such as rubber, plastic, isolation and fibreglass.
Safety Measures and Toxic Values
Styrene’s self-triggered polymerisation is its main danger. This reaction is extremely exothermic and this creates explosion risk. Therefore, it must be used in cool environments and polymerisation inhibitors must be used. It is also extremely flammable and it can cause explosion if it contacts with water.
Styrene is hazardous for human health. It is toxic and also suspected to be carciogenic. There are studies conducted to prove that it causes vision and hearing loss on living beings. It must be stored in well-aired environments and exposure must be avoided.