Cas No: 1310-73-2
EINESC No: 215-185-5
Molecular weight: 39,9971 g/mol
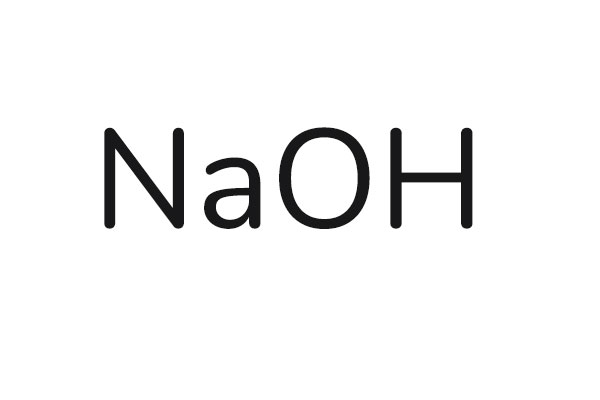
Chemical Formula: NaOH
Chemical Structure:
Physical Properties
General Properties: Nondecane crystal
Odor: Odorless
Intensity: 2.13 g/cm3
Boiling point: 1388 °C
Melting point: 323 °C
Flash point:
Vapor pressure: <2.4 kPa (20 °C)
Refraction index: 1,358 nD
Solubility : 418 g/L (0 °C) (aqueous)
General Properties
Sodium Hydroxide; also known as caustic or caustic soda, is a basic compound. It easily dissolves in water through an exothermic reaction. It is a good corservative for corbone dioxide. It is a commonly used alcali. In 2004, 60 millions of tons of it was produced in the world.
Production
Although it has been produced with different methods throughout history, it is produced abundantly by chlorine alcali process, nowadays.
Applications
Sodium Hyroxide is a strong alcali that is used in industry quite often. It is used in production of sodium salts, detergents and it is also used in organic synthesis as pH stabilizer. It is often used in neutralizing acids as it is a stong alcali.
It is also used as an ingredient in neutralizing drilling systems. It goes through a process that is called low quality crude oil caustic wash. Thus, sulphuric impurities are removed.
In addition to this, it is used in soap making, like potassium hydroxide. It is an effective solvent in unclogging and oil solving. It is also used in cleaning metal surfaces.
It is also used abundantly in pulping paper in paper manufacturing.
It is also used in livestock farming, dissolving atmospheric metals and compounds, reactive for esterification and transesterification, water purificaton facilities, food and construction fields.
Safety Measures and Toxic Values
Sodium Hyraoxide is highly irritant for living tissue. It can easily seperate proteins from lipids on tissue. It may cause permanent injuries if it contacts with eyes. Working with sodium hyroxide without security equipments are not adviced because inhaling its smoke can be dangerous. If it contacts with skin, sluicing is advised. Its LD50 value is 40mg/kg.
It must be contained in impermeable containers when it is stored in large amounts beacuse it is humectant. It must be held in polyethylene, carbone steel, PVC or stainless steel containers because it can damage some metals.