Cas No: 7681-38-1
EINESC No: 231-665-7
Molecular weight: 120.06 g/mol
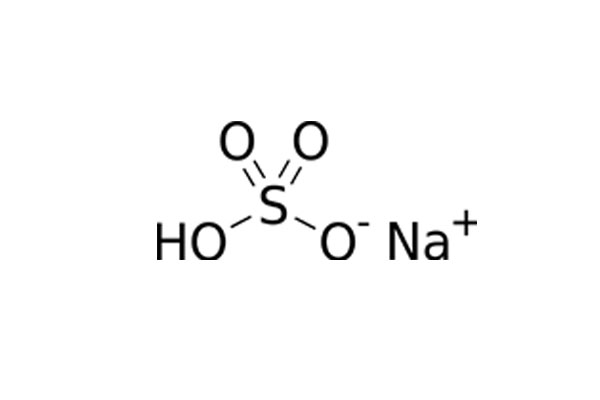
Chemical Formula: NaHSO4
Chemical Structure:
Physical Properties
General Properties: White solid
Odor: Disturbing, similar to rotten eggs
Intensity: 2.742 gr/cm3
Boiling point: 315°C (disintegrated as(Na2S2O7+H2O))
Melting point: 58,5 °C (Monohydrate) 315°C (Dehydrated)
Flash point:
Vapor pressure:
Refraction index:
Solubility : 28,5 gr/100 ml (25°C)
General Properies
Sodium Bisulfate, also known as Sodium Hydrogen Sulfate, is an acid salt. Its anhydrous form is hygroscoptic and in dry form it can be transferred and stored safely. Its aqueous solution is extremely acidic. pH number of its 1 molar solution is less than 1.
Production
Two different methods can be followed for production. First, it can be acquired by reaction of sodium hydroxide and sulphuric acid’s stoichiometric reactions. As a result, sodium bisulfate and water is formed.
Alternatively, sodium chlorine and sulphuric acid are put into reaction in a high temperature. From this reaction, sodium bisulfate and hyrogen chlorine are acquired.
Applications
Sodium Bisulfate is essentially used as pH reducer. It is used after chlorination in swimming pools. It is also used as water’s pH stabilizer in cleaning products.
It is also used as ingredient in animal foods, beverages and sauces.
It is used for preventing cut animal meats from going sour.
It works as polish when used on metal surfaces. Thanks to this property, it is widely used in jewellery business.
It is used in canned foods and cosmetics as anticeptic and disinfectant.
Aside from these, it is also used in textile and leather businesses.
Safety Measures and Toxic Values
Sodium Bisulfate is hazardous for human health. It may cause serious injuries if it is inhaled or contacted the skin. It may be fatal if swallowed.
It releases toxic gas when it contacts with acids or high temperatures.