Cas No: 144-55-8
EINESC No: 205-633-8
Molecular weight 84,0066 g/mol
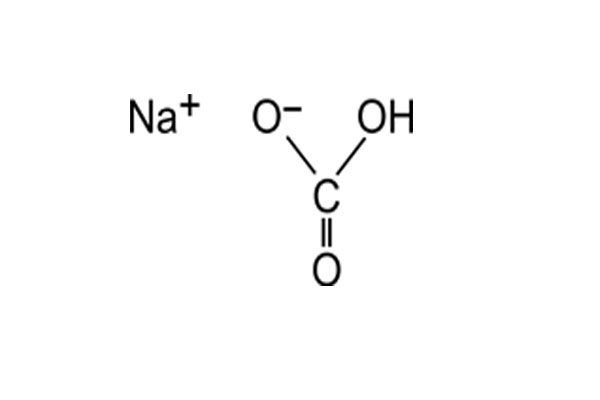
Chemical Formula: NaHCO3
Chemical Structure:
Physical Properties
General Properties: White crystals
Odor: Odorless
Intensity: 2.20 g/cm3
Boiling point: –
Melting point: –
Flash point: –
Vapor pressure: –
Refraction index: –
Solubility : 69 g/L (0 °C)
General Properties
Sodium bicarbonate, commonly known as baking soda, is a sodium salt. It is commonly found as white solid crystals but it can be found as powder. It has a slightly salty and alkaline taste. It is found as nahcolite in nature.
Production
Sodium Bicarbonate is industrially produced from reaction of sodium carbonate and aqueous solution of high intensity carbone dioxide.
Applications
Sodium Bicarbonate has a wide array of usage. Firstly, it is used in baking powder. When heated, it resolves to carbone dioxide and water. This carbone dioxide allows dough to enlarge.
Aside from this, it show a slight alkaline property when it reacts to water, thus it can be used as detergent. Thanks to this property, it can also be used in toothpaste.
It is also commonly used in livestock farming, fire extinguishers, paper, leather, textile and purification of waste water.
Safety Measures and Toxic Values
It is not too harmful when it comes to environment or health. Only, it may cause irritation when it contacts with skin. If it contacts with eyes, it may cause vision loss.