Cas No: 78-93-3
EINESC No: 201-159-0
Molecular weight: 72.107 g/mol
Chemical Formula: C4H8O
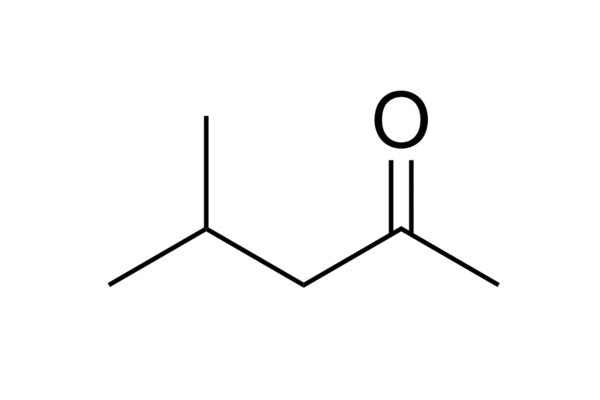
Chemical Structure:
Physical Properties
General Properties: luminous, colorless liquid
Odor: pleasant, fruity
Intensity: 0,8050g/cm3
Boiling point: 79.64 °C
Melting point: −86 °C
Flash point: – 9 °C
Vapor pressure: 78 mmHg (20 °C)
Refraction index: 1,37880 nD
Solubility : 27,5 g/100 mL
Viscosity:
General Properties
Methyl ethyl ketone, or MEK, is known to be a butanone. It is a colorless, liquid, acetone-like ketone that has room temperature. It is partly dissolves in water. It is commonly used in industry. MEK has low toxicity and it is extremely flammable. It is found in fumes of tobacco, wood and engine oil. It is also a natural product that is found in certain fruits, vegetables and other food types.
MEK has solvent properties which are similar to acetone. However, it boils in higher temperatures and it has considerably slower vaporization speed. Unlike acetone, it forms an azeotrope.
Production
Methyl ethyl ketone is produced by oxidation of 2-butanoline. And 2-butanoline hydrogen removing process is started. It is catalyzed by copper, zinc and bronze.
Applications
Methyl ethyl ketone provides rapid drying in dyeing business thanks to its high vaporization speed. It is used as solvent for dye stripping, paper coating, flexography, rubber production and wood varnishes. It is used as a solvent in felt production, cold cleaning and oil vaporizing processes. It is also a starting material for cleaning fluids and other chemicals.
It is used in plastic welding, as well. Although it is mistaken for glue, MEK is a plastic welding agent as it allows plastics to stick to each other by melting them.
Aside from these, MEK also used in polyester resin production as catalyst.
Safety Measures and Toxic Values
Methyl ethyl ketone has low toxicity. However, it is irritant for skin. It may cause serious damages to eyes. Its fume may cause airway diseases.
Methyl ethyl ketone is extremely flammable and it may create explosive mixtures with air. Its peroxide is a much more stronger explosive. It must be kept away from oxidizers. Therefore, it must be stored in tightly closed, cool containers and it must be kept away from fire sources.