Cas No: 7664-39-3
EINESC No: 231-634-8
Molecule weight: 20,01 g/mol
Chemical Formula: HF
Chemical Structure:
Physical Properties
General Properties: Colorless, liquid
Odor: Strong, abrasive
Intensity: 1,15 g/mL3
Boiling point: 19,5 °C
Melting point: -83,6°C
Flash point:-
Vapor pressure: 783 mmHg (20 °C)
Refraction index: 1,00001 nD (25 °C)
Solubility (aqueous) completely miscible
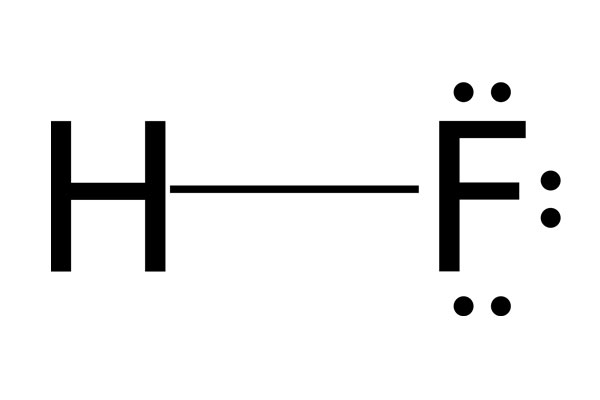
General Propeties
Hydrofluoric is aqueous solution of hydrogene fluoride. Although its boiling point is higher than other hydro halogens, it is lower than room temperature thus, it is sold as aqueous solution form. In high temperatures, it is more stable in pure form. It takes form of sextette or septette groups in low temperatures as hydrogene and fluorine atoms form inter-molecular bonds. In its pure form and highly concentrated solutions, it ionizes by itself and is extremely acidic. In lower concentrations, its acid level decreases but it is still highly abrasive to skin. It is also abrasive to glass therefore, glass containers should not be used.
Production
It is generally acquired from reaction of sulphuric acid and fluorine. 20% of the produced substance is transformed into hexafluorosilic acid. Then, if desired, it can be made more fruitful through hydrolysis.
Applications
It is commonly used in mining, glass and wood businesses. It is a proper acid to melt down silicate based materials such as rocks in natural surfaces. It also plays a vital role in purification of aluminium and uranium. It is the pioneer of organofluorines and it is used in production of chemicals such as teflon and chloroform.
It is also commonly used in petrochemistry and medical fields as it is a very good solvent.
Safety Measures and Toxic Levels
Hydrofluoric acid is extremely dangerous to human health. Inhaling its smoke or making it contact to eyes or skin may cause serious health problems. It vaporizes easily due to its low boiling point and when it is inhaled, it causes lungs to collect water and possibly resulting in death. When it is contacted with skin, it lacerates tissue rapidly and causes serious injuries, it can also lacerate cornea rapidly and cause permanent blindness. Any kind of exposure requires special treatment and meical care. It must be stored in a well-aired place as it is highly volatile. Containers must be closed tightly. The place must be aired when using it.