Cas No: 65-85-0
EINESC No: 200-618-2
Molecular weight: 122,123 g/mol
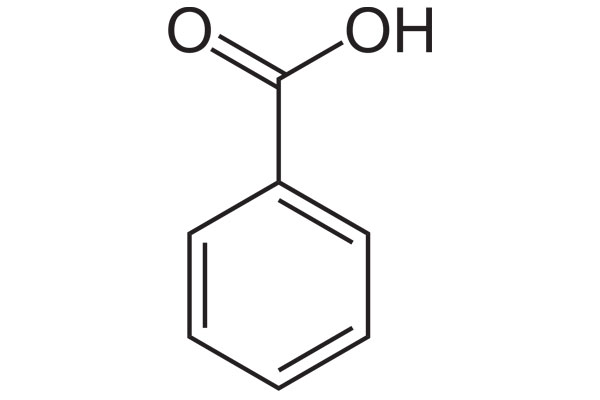
Chemical Formula: C7H6O2
Chemical Structure:
Physical Properties
General Properties: Odorless, crystalline solid
Odor: Slightly pleasant
Intensity: 1,2659 g/cm3
Boiling point: 250 °C
Melting point: 122 °C
Flash point: 121,5 °C
Vapor pressure: 0.16 Pa (25 °C)
Refraction index: 1,5397 (20 °C)
Solubility : 3,44 g/L 25 °C
General Properties
Benzoic Acid, simplest member of the aromatic carboxylic acid family, is a weak acid that is pioneer to synthesis of many important organic compound.
Benzoic acid is a white paillette and crystal-ish solid, it dissolves in water poorly. It is found naturally in many plants. It also works as a byproduct in biosynthesis of many secondary metabolism products.
Benzoic acid dissolves in ethanol completely while partly dissolving in benzane and acetone.
Production
Benzoic acid is produced from toluen in trade business and this process is catalyzed by either cobalt or magnese napththenates. This reaction usually proceeds fruitfully.
Besides, it can easily be produced by oxidation of benzil derivatives such as; benzil alcohol, benzil chlorine, carboxylation or by small amount of bromobenzene grignard reaction.
Applications
Its production mainly focuses on phenol manifacturing. It is used as a primary ingredent for this plastic phenol production. Besides, benzoic acid is a very common food protector and it is used to prevent microbial degradation in many food products from ketchup to beverages. It is usually determined by a country’s legislation on how much benzoic acid should be used. Regarding this topic, there is a concern that benzene, a carcinogenic substance, will form from its reaction with ascorbic acid/vitamin C. Benzoic acid’s solutions are commonly used in treating diseases that are fungus-based such as; foot fungus or alopecia.
It is used as byproduct in fly-repellants, automotive cosmetics and textile business.
Safety Measures and Toxic Values
Benzoic acid is irritant for eyes and skin. Because it is an acid, its contact with eyes must be avoided and exposure to its steam for long periods of time must also be avoided. Otherwise, it may cause serious lung problems.